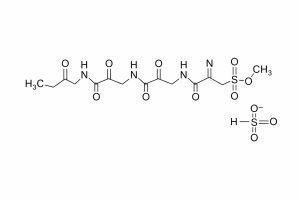
Deferoxamine Mesylate
Chemox Pharma is a leading and a top Deferoxamine Mesylate manufacturer in India, supplying high-quality pharmaceutical-grade APIs to global markets. Deferoxamine Mesylate

Fexofenadine Hydrochloride [Cas No. 153439-40-8] is a non-sedating second generation antihistamine API which is usually used in treatment of seasonal allergic rhinitis and chronic urticaria. Fexofenadine Hydrochloride delivers rapid and targeted relief from allergy symptoms such as itching, runny nose, sneezing as it acts as a selective H1-receptor antagonist.
Fexofenadine Hydrochloride is utilized as oral solid dosage forms for example: tablets, capsules and suspension that helps in providing fast action. It does not cross blood brain barrier and reduces the risk of anticholinergic , sedation and antidopaminergic effects. Fexofenadine Hydrochloride has a high tolerance level and has a good safety profile thus is preferred choice among all types of age group.
Chemox Pharma is a leading manufacturer of Fexofenadine Hydrochloride with its multipurpose ultramodern state-of-the-art facility at Dahej Industrial area of Gujarat, India. Chemox Pharma actively seeks global regulatory approvals and files Drug master files (DMFs). We aim to create high quality products and innovative APIs through continuous R&D following the latest WHO – GMP standards.
| Sr. No. | Description | Application |
|---|---|---|
| 1 | Antihistamine API | Anti-allergic tablet formulations |
| 2 | H1 receptor antagonist drug substance | Allergic rhinitis treatment |
| 3 | Non-sedating antihistamine ingredient | Chronic urticaria therapy |
| 4 | Anti-allergy pharmaceutical API | Respiratory allergy formulations |
| 5 | Second-generation antihistamine | Seasonal allergy medications |
Fexofenadine Hydrochloride is widely used by pharmaceutical manufacturers in oral antihistamine formulations for allergy and urticaria management.
Fexofenadine Hydrochloride API should be handled according to standard pharmaceutical material safety and industrial hygiene procedures. Appropriate protective measures must be followed during handling and processing of the antihistamine active pharmaceutical ingredient in GMP manufacturing environments.
Use appropriate personal protective equipment (PPE)
Avoid inhalation of powder and contact with skin or eyes
Handle in controlled pharmaceutical manufacturing areas
Follow GMP material handling procedures
Refer to Safety Data Sheet (SDS) for detailed safety guidance
This API is intended exclusively for pharmaceutical manufacturing and formulation use.
Fexofenadine Hydrochloride is a selective peripheral H1 histamine receptor antagonist that blocks the effects of histamine released during allergic reactions. By inhibiting histamine binding at H1 receptors in respiratory and dermal tissues, it reduces symptoms such as sneezing, itching, rhinorrhea, and urticaria. Its limited penetration into the central nervous system results in minimal sedation.
Handle in approved pharmaceutical manufacturing facilities
Avoid exposure without protective equipment
Prevent dust generation and inhalation
Follow GMP containment procedures
Dispose according to regulatory guidelines
Store in tightly closed containers
Protect from moisture and light
Maintain controlled room temperature
Keep in designated API storage areas
Follow GMP warehouse practices
Fexofenadine Hydrochloride API is used in pharmaceutical formulations for the treatment of allergic rhinitis and chronic urticaria.
Yes, Chemox Pharma is a WHO-GMP certified Fexofenadine Hydrochloride API manufacturer and supplier in India serving global pharmaceutical companies.
The CAS number of Fexofenadine Hydrochloride API is 153439-40-8.
Fexofenadine Hydrochloride belongs to the second-generation antihistamine (H1 receptor antagonist) class used in allergy treatment.
It should be stored in tightly closed containers at controlled room temperature, protected from moisture and light, following pharmaceutical storage guidelines.

Chemox Pharma is a leading and a top Deferoxamine Mesylate manufacturer in India, supplying high-quality pharmaceutical-grade APIs to global markets. Deferoxamine Mesylate
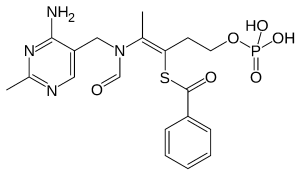
Chemical Name: Benfotiamine (S-benzoylthiamine O-monophosphate) CAS Number: 22457-89-2 Molecular Formula: C₁₉H₂₃N₄O₆PS Molecular Weight: 466.45 g/mol Therapeutic Uses: Diabetic neuropathy, neuroprotection, nerve &
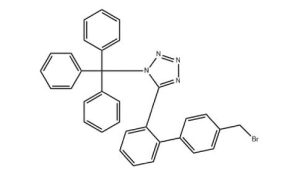
API name/end use: Sartan family Cas number: 124750-51-2 Molecular formula: C33H25BrN4 Molecular Weight: 557.48 Appearance: Solid Powder Storage: Room Temperature (Recommended in