Introduction
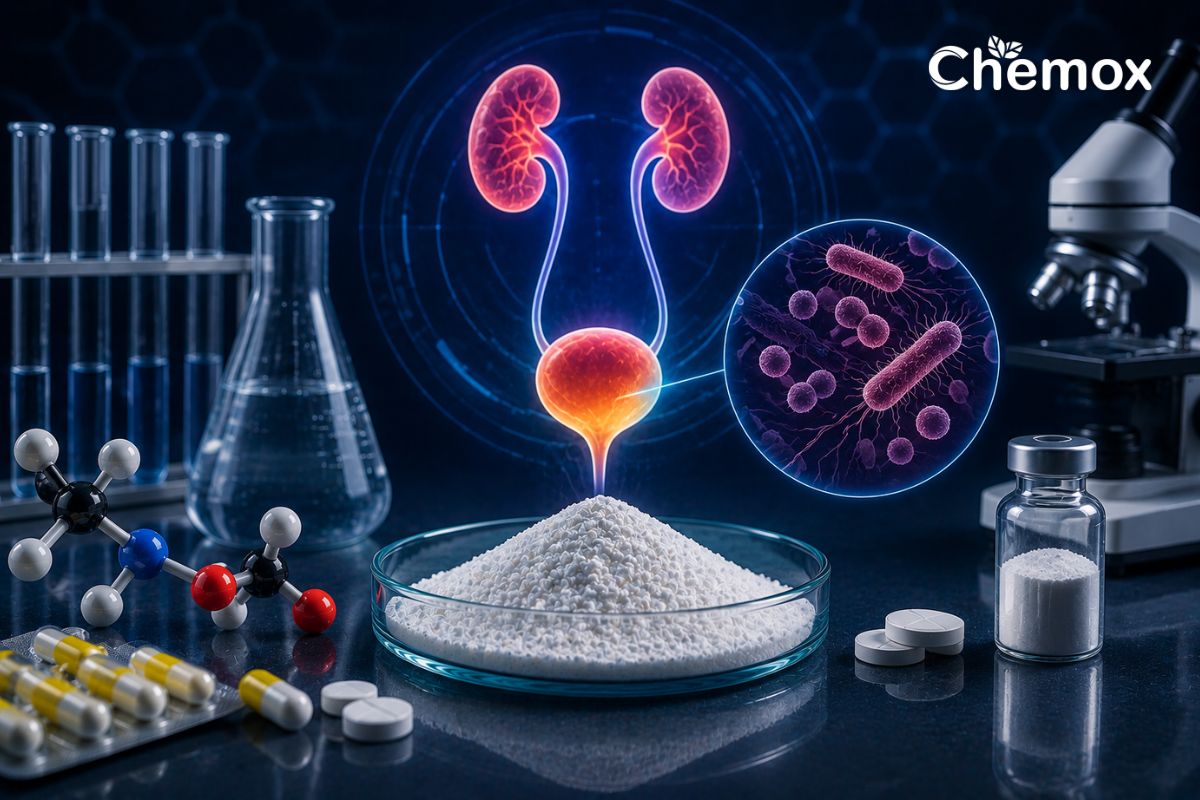
Urinary Tract Infections (UTIs) represent a significant global health challenge, affecting millions annually. In the ongoing battle against these common bacterial infections, Nitrofurantoin Active Pharmaceutical Ingredient (API) stands as a foundational and highly effective treatment. Its enduring efficacy and favorable resistance profile make it a critical component in the pharmaceutical arsenal against UTIs. This article delves into the therapeutic importance of Nitrofurantoin API, the stringent manufacturing controls essential for producing a high-purity and effective API, and highlights Chemox Pharma’s expertise in antibacterial API production.
The Therapeutic Importance of Nitrofurantoin in UTI Treatment
Nitrofurantoin, a synthetic nitrofuran derivative, has been a cornerstone in UTI treatment since its introduction in 1953 . Its unique mechanism of action sets it apart from many other antibiotics. Instead of targeting a single bacterial pathway, Nitrofurantoin works by inhibiting multiple bacterial enzyme systems, including those involved in DNA and RNA synthesis, protein synthesis, and aerobic energy metabolism . This multi-targeted approach contributes to its sustained efficacy and a lower propensity for resistance development compared to other classes of antibiotics, such as fluoroquinolones or sulfonamide.
Crucially, Nitrofurantoin is concentrated in the urine, making it exceptionally effective for treating uncomplicated lower UTIs. This localized action minimizes systemic exposure and potential side effects, further solidifying its role as a first-line treatment option. The World Health Organization (WHO) recognizes Nitrofurantoin as an essential medicine, underscoring its critical role in global public health .
Manufacturing Excellence: Ensuring High-Purity Nitrofurantoin API
The effectiveness and safety of any pharmaceutical product are directly linked to the quality of its active pharmaceutical ingredient. For Nitrofurantoin API, stringent manufacturing controls are paramount to ensure high purity, consistent quality, and optimal therapeutic performance. Manufacturers must adhere to global regulatory standards, including WHO-GMP (Good Manufacturing Practices), and comply with pharmacopoeial requirements such as IP (Indian Pharmacopoeia), BP (British Pharmacopoeia), EP (European Pharmacopoeia), and USP (United States Pharmacopoeia) .
Key considerations in Nitrofurantoin API manufacturing include:
- Purity Standards: Achieving and maintaining a high purity level, typically exceeding 99%, is crucial to minimize impurities that could affect drug safety and efficacy.
- Particle Size Distribution (PSD): Nitrofurantoin is often available in different physical forms, each with specific particle size characteristics that influence its dissolution rate, bioavailability, and gastrointestinal tolerance. These forms include:
- Nitrofurantoin Anhydrous: Ideal for standard tablet and capsule formulations.
- Nitrofurantoin Monohydrate: Often used in combination with macrocrystals for sustained-release formulations, offering smaller particle sizes and higher solubility for rapid dissolution .
- Nitrofurantoin Macrocrystals: Specifically engineered with larger particles for better gastrointestinal tolerance and controlled absorption, reducing common side effects like nausea .
Precise control over the crystallization process and subsequent particle size reduction techniques are vital to produce these distinct forms, ensuring they meet the specific needs of various pharmaceutical formulations.
Chemox Pharma’s Expertise in Antibacterial API Production
As a leading Nitrofurantoin API manufacturer, Chemox Pharma is committed to delivering high-quality, compliant ingredients to the global pharmaceutical industry. Our state-of-the-art manufacturing facility in Dahej, Gujarat, operates under strict WHO-GMP guidelines, employing advanced technology and rigorous quality control measures at every stage of production .
Chemox Pharma’s commitment to excellence is demonstrated through:
- WHO-GMP Certified Production: Our adherence to global GMP standards ensures that every batch of Nitrofurantoin API is produced under controlled conditions, guaranteeing consistent quality and purity.
- Comprehensive Regulatory Support: We provide extensive support for regulatory submissions, including Drug Master File (DMF) filings and complete documentation for IP, BP, EP, and USP grades. This streamlines the approval process for our partners’ finished dosage forms in diverse international markets .
- Diverse Product Forms: Understanding the varied formulation needs, Chemox Pharma offers Nitrofurantoin in its Anhydrous, Monohydrate, and Macrocrystalline forms, providing flexibility and tailored solutions for different drug delivery systems.
- Strategic Location and Supply Chain: Situated in India’s pharmaceutical hub, Chemox Pharma benefits from robust logistics and a well-established supply chain, enabling competitive pricing and timely delivery to over 90 countries worldwide .
Our scientific expertise, combined with decades of experience in antibacterial API production, positions Chemox Pharma as a trusted partner for pharmaceutical formulators and procurement teams seeking reliable and high-purity Nitrofurantoin API.
Conclusion
Nitrofurantoin API remains an indispensable treatment for urinary tract infections, valued for its efficacy and low resistance profile. The successful formulation of Nitrofurantoin-based drugs hinges on the quality and purity of the API, necessitating stringent manufacturing controls and adherence to global regulatory standards. Chemox Pharma stands at the forefront of this critical field, offering high-purity Nitrofurantoin API backed by WHO-GMP certified production, comprehensive regulatory support, and a commitment to manufacturing excellence. Partnering with a reputable manufacturer like Chemox Pharma ensures the integrity and effectiveness of UTI treatments, ultimately benefiting patients worldwide.