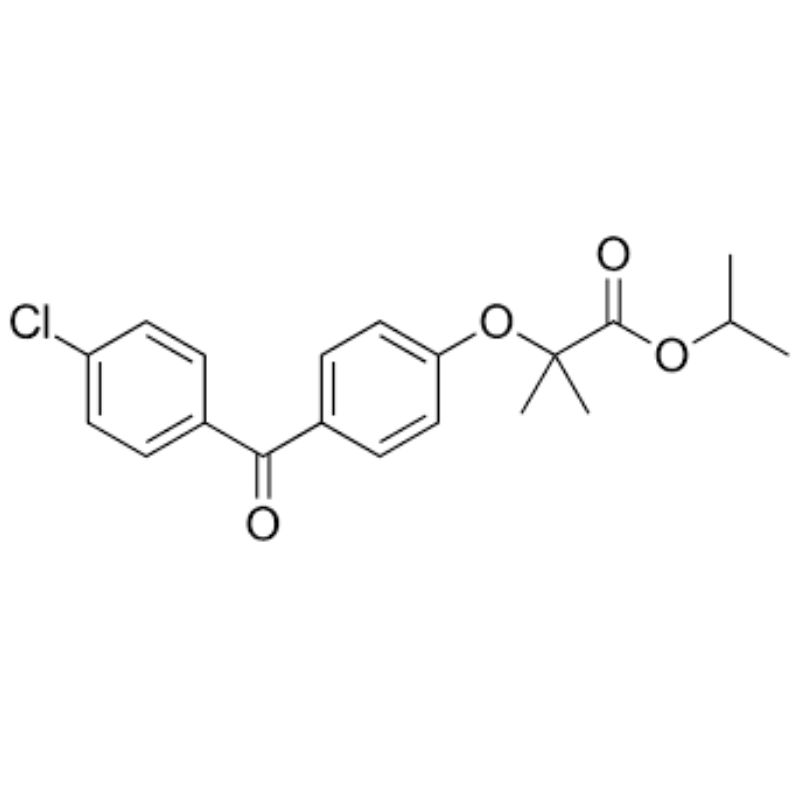
Product Specifications
Fenofibrate API should be handled according to standard pharmaceutical material safety and industrial hygiene procedures. Appropriate protective measures must be followed during handling and processing of the cardiovascular active pharmaceutical ingredient in GMP manufacturing environments.
-
Use appropriate personal protective equipment (PPE)
-
Avoid inhalation of powder and contact with skin or eyes
-
Handle in controlled pharmaceutical manufacturing areas
-
Follow GMP material handling procedures
-
Refer to Safety Data Sheet (SDS) for detailed safety guidance
This API is intended exclusively for pharmaceutical manufacturing and formulation use.